Back to all news and events

22 January 2019
PQ Bypass Announces Appointment of Stanton J. Rowe to Board of Directors
Meditrial Europe is one of the first and few CROs awarded ISO 9001:2015 certification, confirming its leadership in clinical research and digital solutions for the medical device industry.
Read more

3 April 2018
Meditrial Europe awarded ISO 9001:2015 Certification
Meditrial Europe is one of the first and few CROs awarded ISO 9001:2015 certification, confirming its leadership in clinical research and digital solutions for the medical device industry.
Read more

30 October 2017
HighLife closes €12.3 million financing round led by Sofinnova Partners
This financing allows for the acceleration of the company’s clinical and R&D program
Read more

11 October 2017
BioVentrix Announces First Patient Enrolled in the U.S. arm of IDE Study of the Revivent TC™ TransCatheter Ventricular Enhancement Treatment
The trial is designed to demonstrate the safety and effectiveness of the Revivent TC™ TransCatheter Ventricular Enhancement System
Read more

5 October 2017
CARMAT expands its PIVOTAL study internationally, in line with its clinical strategy and CE marking process
Approval granted to perform implants in Kazakhstan, leading country in heart device trials for the European market
Read more

2 October 2017
Boston Scientific Announces Agreement to Acquire Apama Medical
Acquisition to Strengthen Company’s Electrophysiology Portfolio and Expand into “Single-Shot” Balloon Pulmonary Vein Isolation Treatments for Patients with Atrial Fibrillation
Read more

1 September 2017
CardiAQ Valve Tech vs Neovasc : Ruling from Appeals Court on TIARA patent family
Appeal Court upholds lower court decisions but denies injunction
Read more

15 March 2017
New AHA/ACC guideline on valvular heart disease management
On March 15, 2017 the AHA/ACC have released a focused update of guidelines for the treatment of patients with valvular heart disease.
Read more
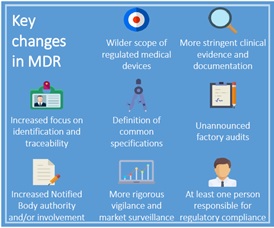
13 March 2017
European council unanimously adopts MDR and IVDR, introducing deep changes in world’s second-largest device market.
The European Union new rules governing medical device and in vitro diagnostics (IVDs) are one step away from final approval.
Read more

6 March 2017
EU Finalizes New Medical Device Regulations (MDR) which update the regulatory framework for the marketing of devices and IVDs in Europe
EU Council will vote the MDR adoption on March 7, 2017.
Read more








